Self-organized bronchoconstriction, tipping points, and ventilation defects in asthma
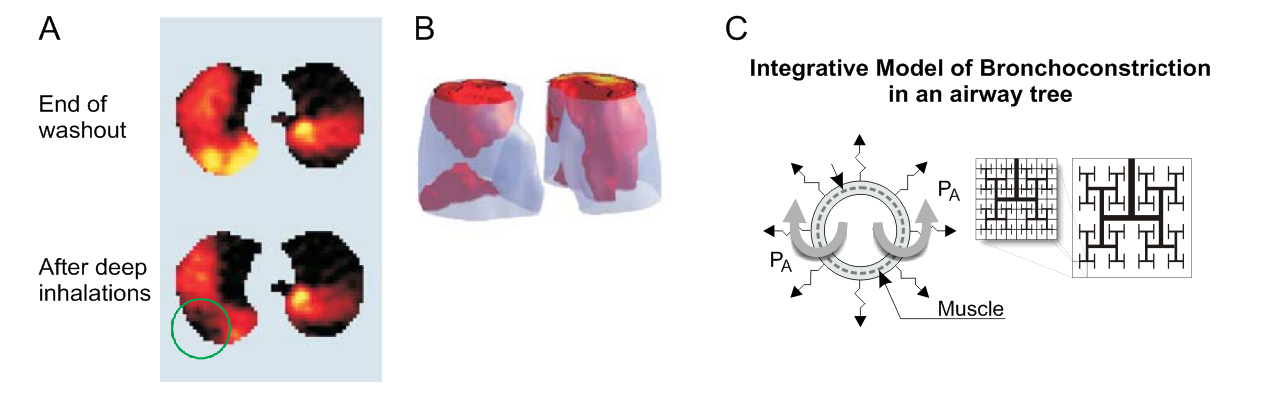
Reversible bronchoconstriction is a key characteristic of asthma and it had been suspected in the past that bronchoconstriction causes some heterogeneity in ventilation. However, using our 13NN-V/Q imaging we have shown that bronchoconstriction results in major regional ventilation defects (VDefs) rather than randomly scattered heterogeneity in ventilation (JG Venegas et al. Nature https://doi.org/bxv5g6; 2005). Remarkably, computational modeling of bronchoconstriction in an airway tree demonstrated that positive feedback in airway behavior can explain the avalanche-like emergence of self-organized clustering of severe airway constriction causing VDefs (ref). Additionally, the computational model can explain a significant number of other non-trivial phenomena of airway behavior (T Winkler et al. JAP https://doi.org/bthhxm; 2007).
To explain the key mechanism of the feedback causing VDefs one can use a single airway bifurcation for simplicity: a lower airflow in one daughter airway results in lower parenchymal tethering leading to an increase in smooth muscle constriction and airway resistance (Fig. 1). When airway narrowing reaches a tipping point triggering this positive feedback, it results in an avalanche-like constriction of the airway. In contrast, the higher airflow in the other airway results in higher regional tethering forces preventing further constriction. An important aspect of this positive feedback causing VDefs is that it occurs only beyond a tipping point in airway narrowing, also referred to as a critical point, triggering the feedback. Thus, minor stimuli of the airway smooth muscle do not lead to VDefs.
Simulation of an asthma attack in the computational model
Our computational model was built to capture the dynamic interactions among airways in response to a stimulus of airway smooth muscle, tidal breathing, and other factors. Visualizing and quantifying the dynamic changes in the airway tree over time has provided unique insights into the complex behavior of airways in asthma. In principle, the self-organized clustering in bronchoconstriction is an emergent phenomenon like starling murmurations or phase separation in physical and biological systems.
Perfusion
Interestingly, bronchoconstriction causes not only VDefs but also regional hypoperfusion: the blood flow inside of VDefs is lower than the blood flow outside (RS Harris et al. AJRCCM doi.org/bkktz8; 2006). We had hypothesized that the obstruction of airflow in VDefs result in hypoxia and, thus, hypoxic vasoconstriction inside of VDefs. However, a detailed investigation using 13NN ventilation and perfusion imaging allowing the estimation of the alveolar oxygen and carbon dioxide partial pressure showed that hypoxic and hypercapnic vasoconstriction cannot fully account for the regional decrease in perfusion (VJ Kelly et al. AJRCCM doi.org/gb4sq5; 2017, VJ Kelly et al. AJRCCM doi.org/gcvm; 2017).
Selected papers
-
Nature 2005;434(7034):777-82. doi: 10.1038/nature03490.
Self-organized patchiness in asthma as a prelude to catastrophic shifts. Jose G Venegas, Tilo Winkler, Guido Musch, Marcos F Vidal Melo, Dominick Layfield, Nora Tgavalekos, Alan J Fischman, Ronald J Callahan, Giacomo Bellani, R Scott Harris. -
J Appl Physiol. 2007;103(2):655-63. doi: 10.1152/japplphysiol.00041.2007.
Complex airway behavior and paradoxical responses to bronchoprovocation. Tilo Winkler, Jose G Venegas. -
Am J Respir Crit Care Med. 2017;196(7):834-844. doi: 10.1164/rccm.201612-2438OC.
Hypoxic Pulmonary Vasoconstriction Does Not Explain All Regional Perfusion Redistribution in Asthma. Vanessa J Kelly, Kathryn A Hibbert, Puja Kohli, Mamary Kone, Elliot E Greenblatt, Jose G Venegas, Tilo Winkler, R Scott Harris.





